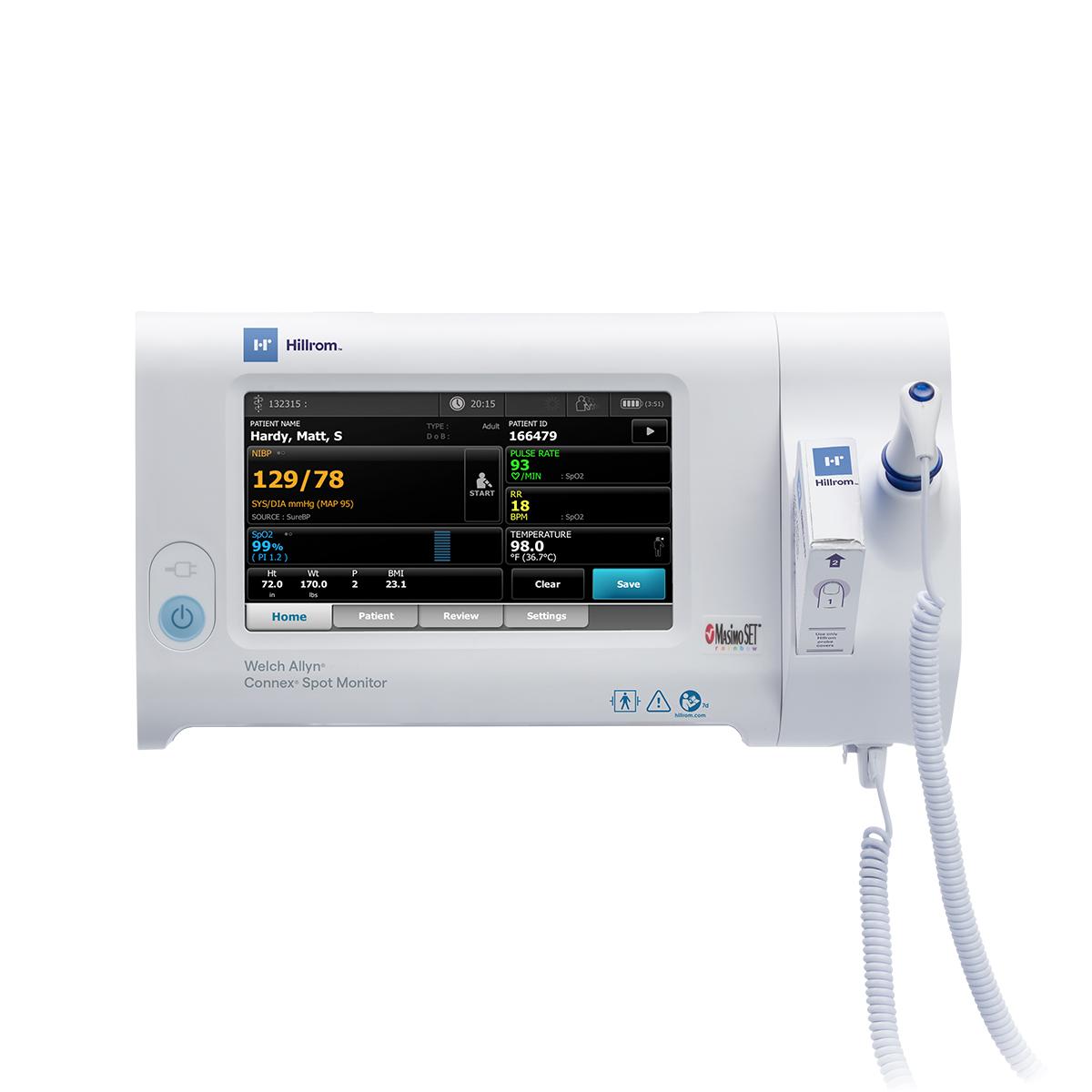
Features
Designed with Patient Outcomes in Mind
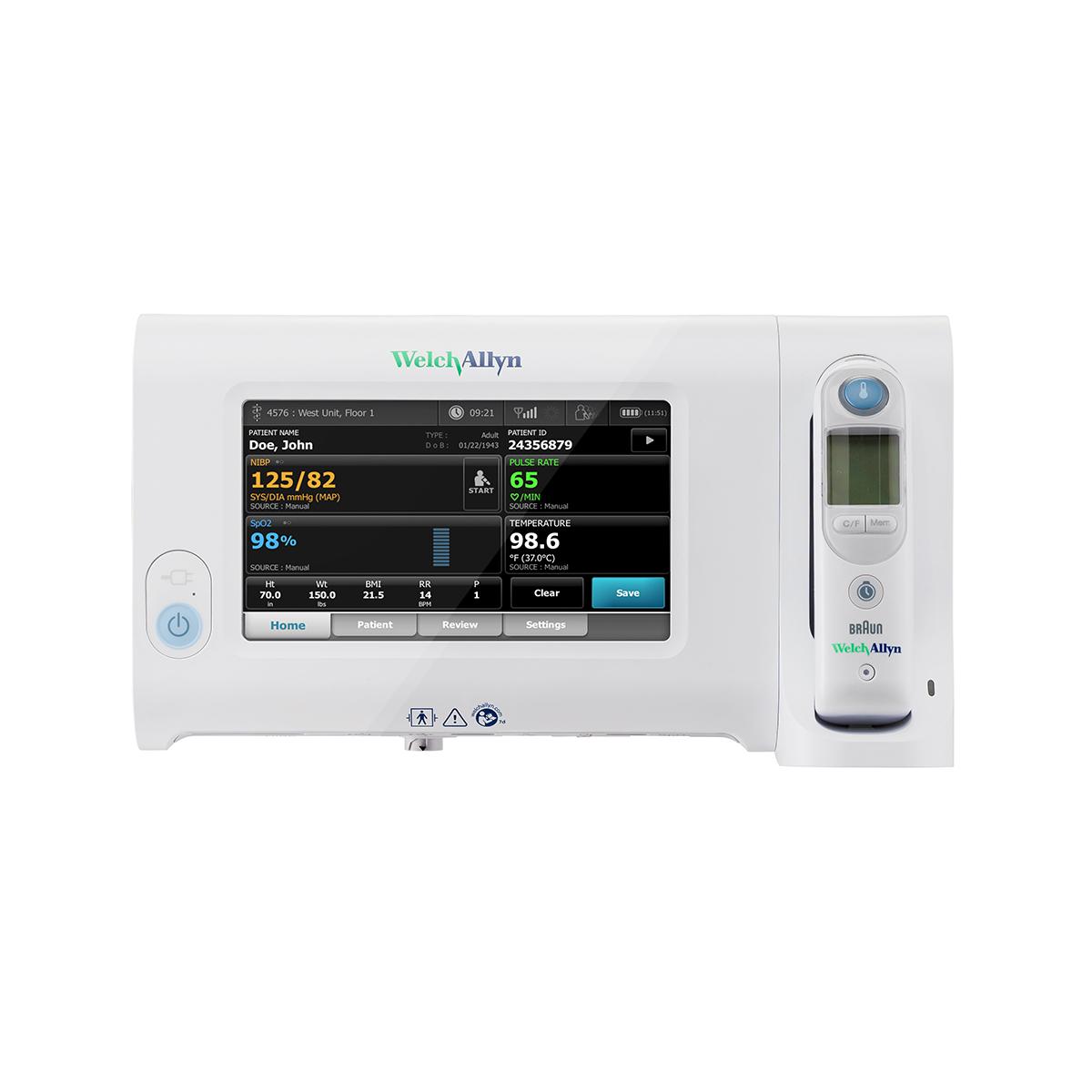
- Measure blood pressure in just 15 seconds with SureBP® technology
- Help ensure accurate hypertension detection with BP averaging
- Choose from SureTemp® Plus and/or Braun ThermoScan® PRO 6000 thermometry
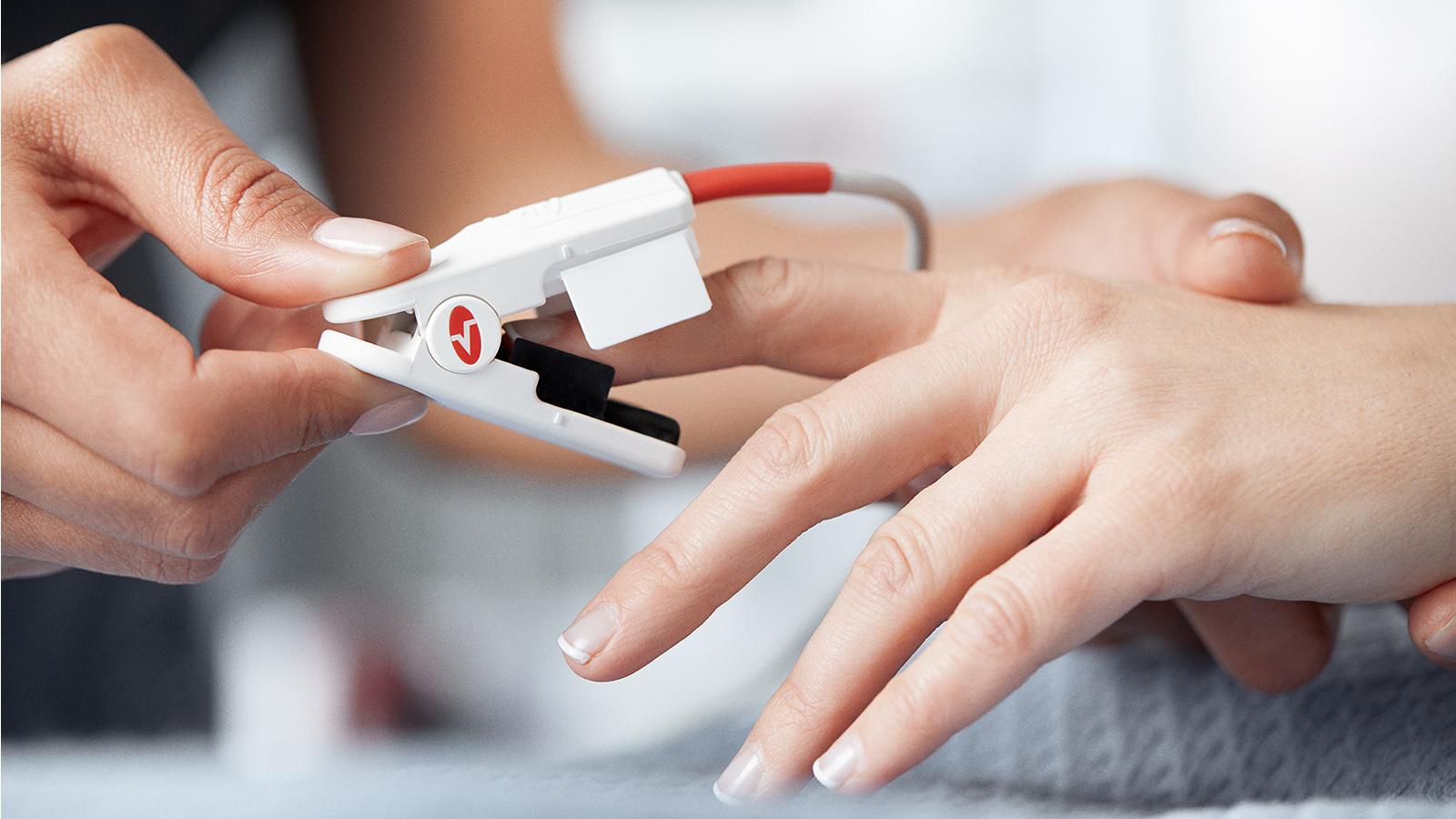
- Capture pulse oximetry with Masimo®, Nellcor® or Nonin® SpO2
- Spot-check respiration rate with the Masimo® RRp® technology
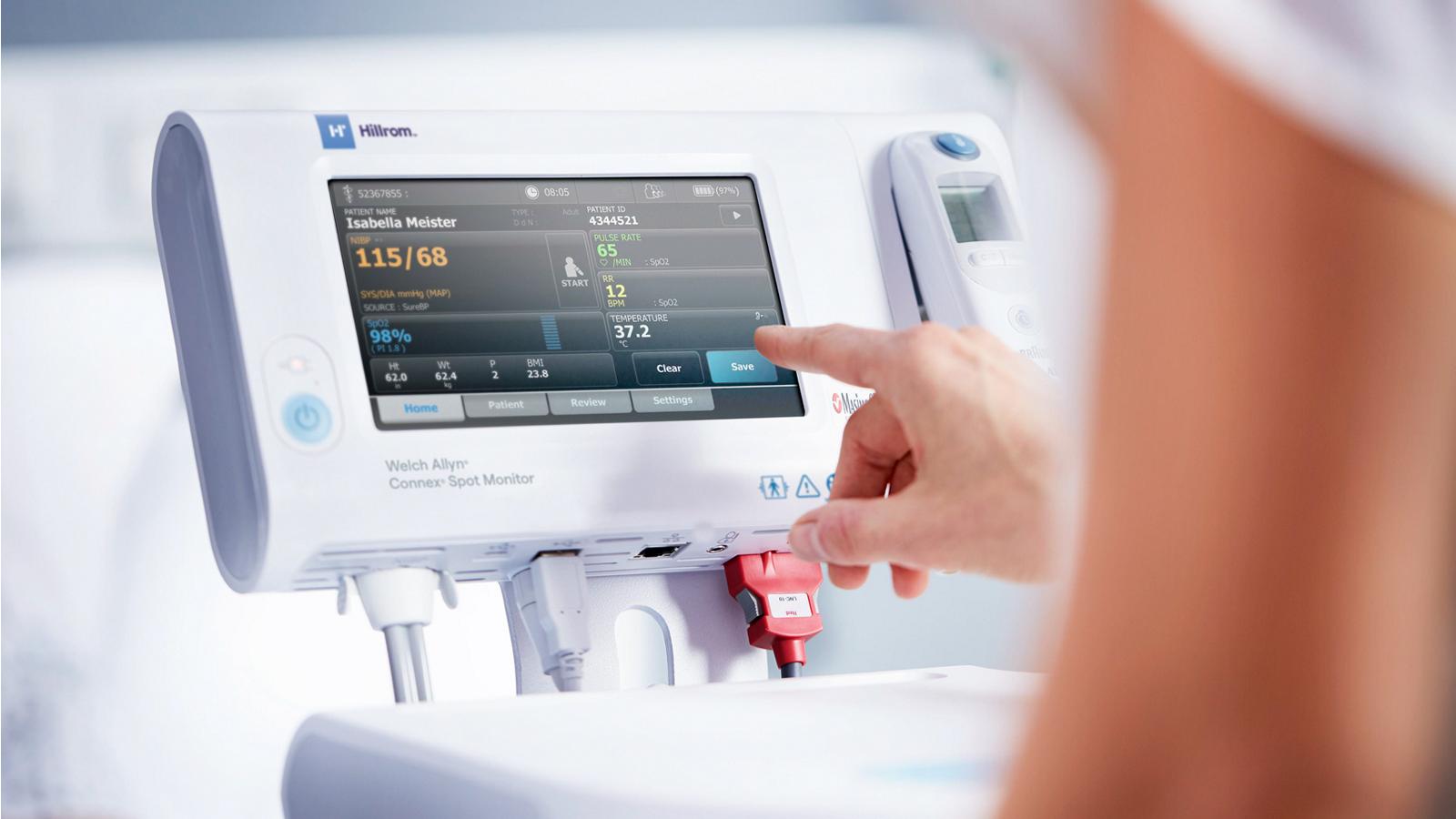
- Connect to your EMR with WiFi®, Ethernet
- Quickly and securely access a device with single sign-on (including Imprivata® integration)
- Help identify signs of patient deterioration with automated Early Warning Scores
- Customize your workflow to document patient observations and vitals modifiers
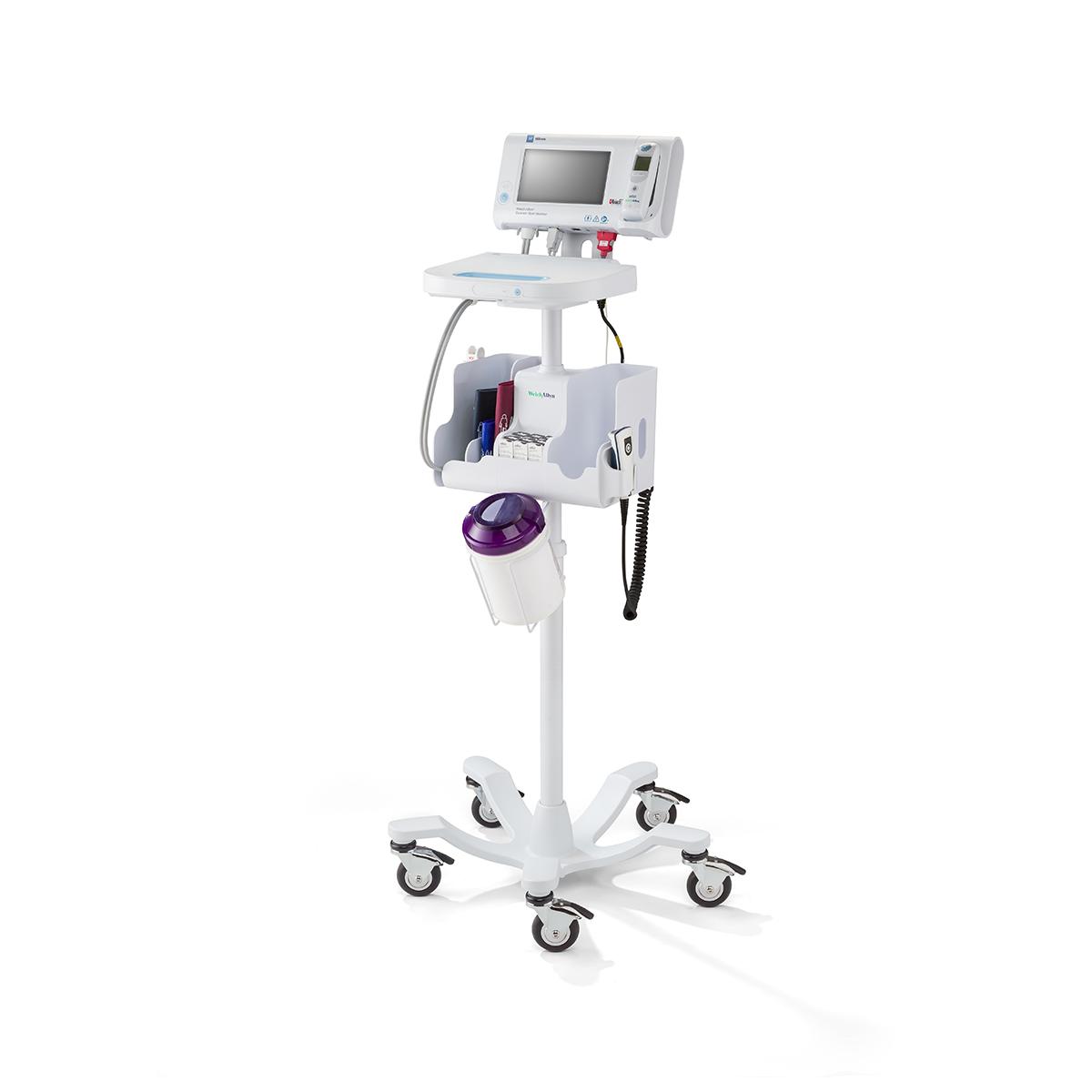
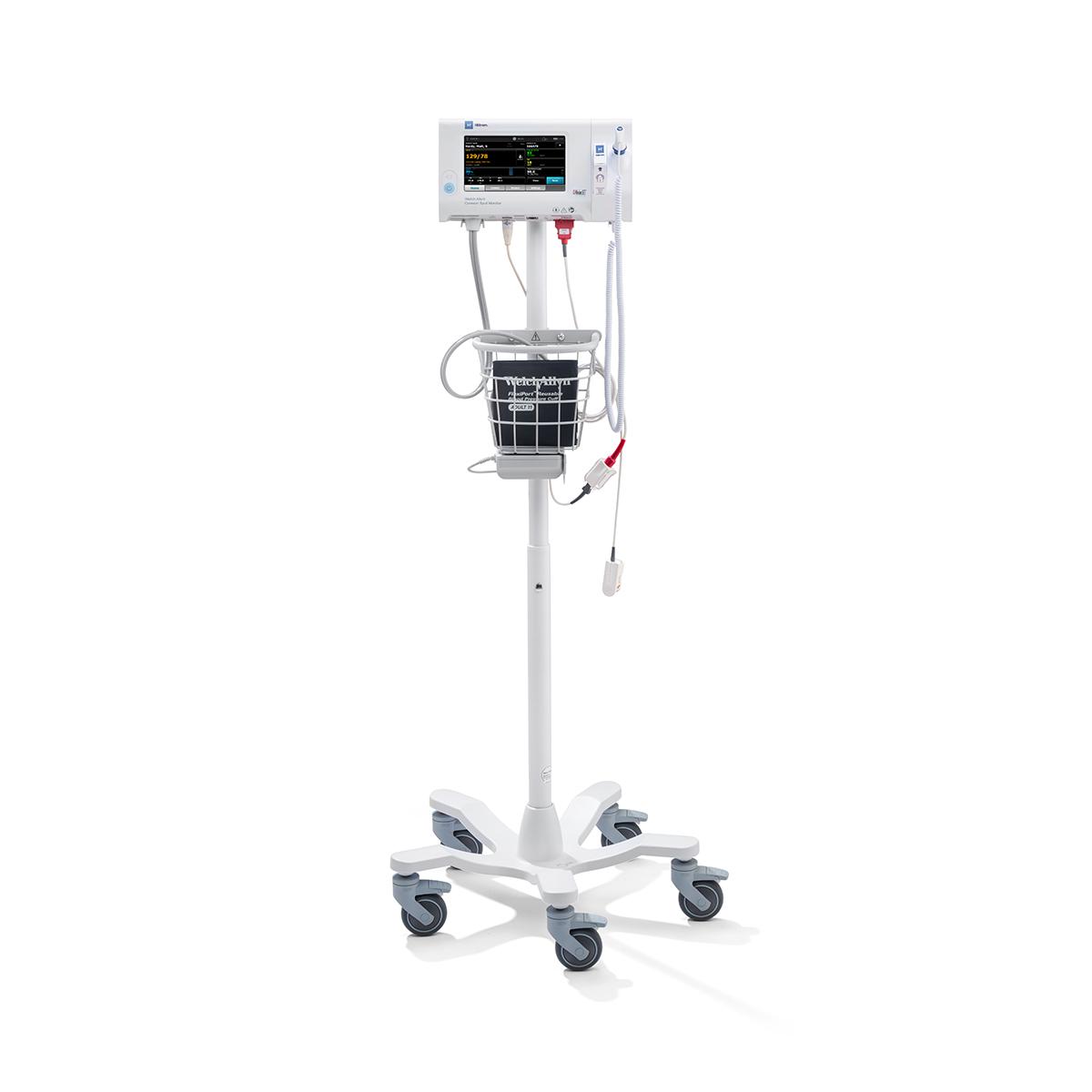
- Meet your workflow needs with mobile stands, wall mounts and integrated exam systems
- Secure patient information with data encryption and additional security features approved by the U.S. Department of Defense (DoD)

Simple
Secure
Connected
Accurate

Are you manually assessing respiration rate?
Spot check respiration rate is now at your fingertips. Automate your workflow to standardize protocols and avoid transcription errors by adding the Masimo® RRp® sensor to your Connex Spot Monitor.
Redefining the Point of Care
Got a Minute?
Education & Documentation
Get in the know to get the most value out of your solution.